7.2 - Protons, Neutrons, Electrons
So we know that the atom is more complicated than people usually picture. There are models of the atom that we will use that will allow us to explain observations in chemistry. We define the atom as something that is the smallest form of an element that still retains all the properties of that element. A gold atom is different than an aluminum atom. The parts that make up atoms are the same though. Just like wood, brick and glass can combine to make a house, they can also combine to make a factory, or a store, or a restaurant. The things that make up any atom are protons, neutrons and electrons. They have different properties that, when combined, make up all the matter in the universe.
The Nucleus
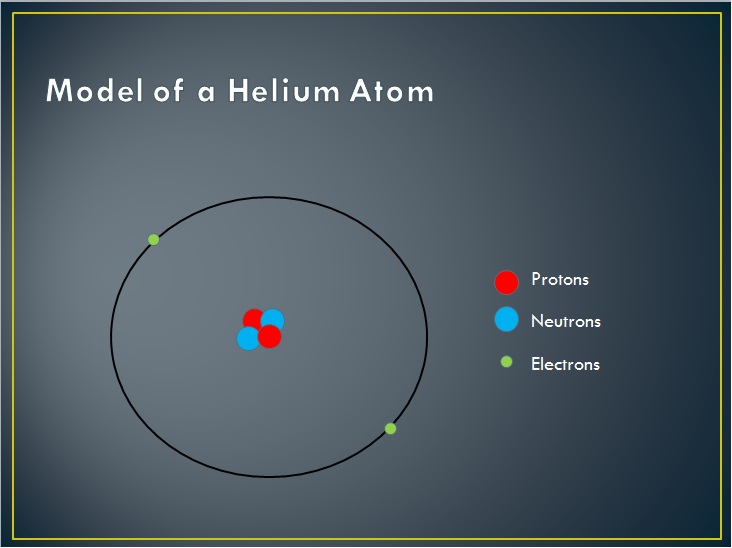
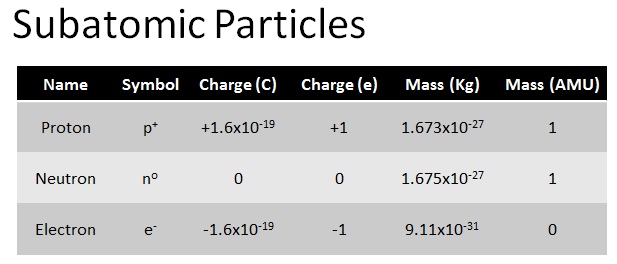
Protons and neutrons are found in the nucleus of the atom. They are in that very tiny, positively charged area at the center of the atom. They have just about the same mass (1.67 X 10^-27 Kg). The proton has a positive electric charge (1.6 X 10^-19 C) while the neutron has no electric charge at all.
While the number of neutrons in the nucleus can vary, the number of protons cannot. It is the number of protons that identifies what an element is. As an example, if one had an atom of carbon, it would have six protons. If there were seven, it would be nitrogen. If there were five, it would be boron. Carbon, and only carbon, has six protons. The number of neutrons varies. Carbon normally has six neutrons. If it had seven, it would still be carbon. There might be some slight differences between it and the other carbon atom, but nothing drastic.
Protons and neutrons are found in the nucleus of the atom. They are in that very tiny, positively charged area at the center of the atom. They have just about the same mass (1.67 X 10^-27 Kg). The proton has a positive electric charge (1.6 X 10^-19 C) while the neutron has no electric charge at all.
While the number of neutrons in the nucleus can vary, the number of protons cannot. It is the number of protons that identifies what an element is. As an example, if one had an atom of carbon, it would have six protons. If there were seven, it would be nitrogen. If there were five, it would be boron. Carbon, and only carbon, has six protons. The number of neutrons varies. Carbon normally has six neutrons. If it had seven, it would still be carbon. There might be some slight differences between it and the other carbon atom, but nothing drastic.
The Electron Orbitals
Electrons do not exist in the nucleus of the atom, they are found in orbitals surrounding the nucleus. The orbitals can also be called electron clouds or energy levels. As you have read, their pattern is very precise- 2, 8, 8, 18, 18, 32, 32. Each orbital must be filled before an electron will go to a higher energy level. Electrons are not nearly as massive as protons and neutrons. They are almost 2000 times less massive (9.11 X 10^-31 Kg).
Electrons do not exist in the nucleus of the atom, they are found in orbitals surrounding the nucleus. The orbitals can also be called electron clouds or energy levels. As you have read, their pattern is very precise- 2, 8, 8, 18, 18, 32, 32. Each orbital must be filled before an electron will go to a higher energy level. Electrons are not nearly as massive as protons and neutrons. They are almost 2000 times less massive (9.11 X 10^-31 Kg).
Watch this video!
7.2 Example Problem
7.2 Review Questions
| 7.2_review_questions.pdf | |
| File Size: | 99 kb |
| File Type: | |