9.5 - Molecular Shapes
Let’s look again at the LDS of different molecules. If we sketch the molecules that are made between hydrogen and the members of the second period, they look like this:
The lines represent bonding pairs of electrons. These are nice, two-dimensional models of the molecules, but we live in a three dimensional world. These diagrams do not represent the shape of the molecule well.
The shapes are determined by the Valence Shell Electron Pair Repulsion Theory (VSEPR Theory). This is a theory that states that the electron pairs around a central atom will get as far from each other as possible, thus determining the shape of the molecule. Let’s look at them one at a time:
The shapes are determined by the Valence Shell Electron Pair Repulsion Theory (VSEPR Theory). This is a theory that states that the electron pairs around a central atom will get as far from each other as possible, thus determining the shape of the molecule. Let’s look at them one at a time:
Alkalai Metals and Halogens
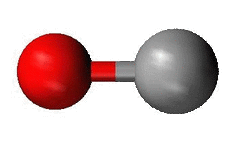
Alkali metals and halogens only have one bonding site. This means that if they are the central atom, the shape of the molecule will be a line. This shape is called linear.
Alkalai Earths
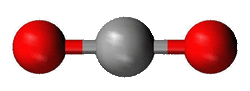
Alkaline earths have two bonding sites. There are no other pairs of electrons around them, so the two bonds get 180° from each other. This is also a linear shape.
Boron Family
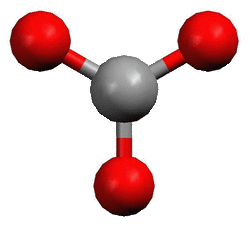
Members of the boron family have three bonding sites with no other pairs of electrons around them. The furthest those three bonds an get from each other is 120°. This shape is called planar trigonal.
Carbon Family
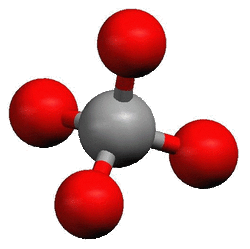
Although, in a two-dimensional drawing, it looks like the four bonds around carbon are as far from each other as they can be, they are not. The bonds shown in LDS format are 90° from each other. When seen three-dimensionally, they get 105° from each other. This is called tetrahedral.
Nitrogen Family
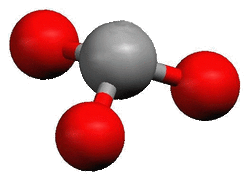
Although members of the Nitrogen Family have three bonding sites like the Boron Family, they do not make the same shape. Members of the Nitrogen Family already have a pair of unbonded electrons. This pair actually pushes the other three bonds away from the planar trigonal shape. The shape they make is pyramidal.
Oxygen Family
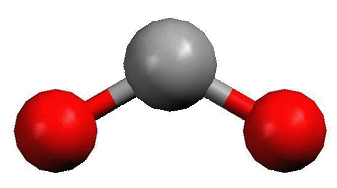
They do have two bonding sites like the Alkaline Earths, but they also have two pairs of unbonded electrons. These pairs push the other two bonds into a bent line shape.
Review the file below and then try the Review Questions.
| 09.3_molecular_shapes.ppt | |
| File Size: | 569 kb |
| File Type: | ppt |
| 9.5_review_questions.pdf | |
| File Size: | 97 kb |
| File Type: | |