7.6 - Ions
Like the opposite ends of a magnet, an electromagnetic force attracts the electrons to the nucleus because the protons reside there. Protons and electrons are oppositely charged and opposites attract. The electrons, being all the same type of charge, repel each other so we have the complex set up of different orbitals surrounding the nucleus. The further away an electron is from the nucleus, the smaller the attraction between it and the protons.
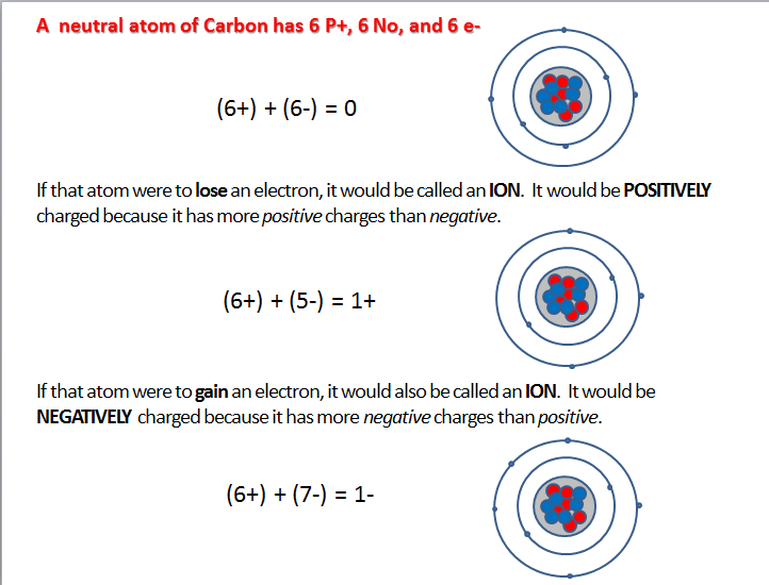
Electrons can be pulled off of an atom. Electrons can also be added to an atom. We usually picture atoms as having the same number of protons as electrons. When that is true the atom is called electrically neutral.
When an atom does not have the same number of protons as it does electrons, that atom is called an ion. Almost all atoms can lose or gain electrons. That is how they react and combine with other atoms to make molecules. The electric charge of an ion is determined by how many protons and electrons it has.
As an example, iron normally has 26 protons and 26 electrons. If it were to lose or gain any protons it would not be iron, so we only have to concern ourselves with the electrons. When iron loses one electron, it would be called Fe1+. It has 26 p+ and only 25 e- . That gives an overall charge of (1+). If iron were to gain one electron, it would be Fe1-. It has 26 p+ and now 27 e- . That gives an overall charge of (1-).
An ion’s charge is determined by the sum of its protons’ and electrons’ charges. When an atom loses electrons, it becomes a positive ion. When an atom gains electrons, it becomes a negative ion.
Electrons can be pulled off of an atom. Electrons can also be added to an atom. We usually picture atoms as having the same number of protons as electrons. When that is true the atom is called electrically neutral.
When an atom does not have the same number of protons as it does electrons, that atom is called an ion. Almost all atoms can lose or gain electrons. That is how they react and combine with other atoms to make molecules. The electric charge of an ion is determined by how many protons and electrons it has.
As an example, iron normally has 26 protons and 26 electrons. If it were to lose or gain any protons it would not be iron, so we only have to concern ourselves with the electrons. When iron loses one electron, it would be called Fe1+. It has 26 p+ and only 25 e- . That gives an overall charge of (1+). If iron were to gain one electron, it would be Fe1-. It has 26 p+ and now 27 e- . That gives an overall charge of (1-).
An ion’s charge is determined by the sum of its protons’ and electrons’ charges. When an atom loses electrons, it becomes a positive ion. When an atom gains electrons, it becomes a negative ion.
7.6 Review Questions
| 7.6_review_questions.pdf | |
| File Size: | 101 kb |
| File Type: | |