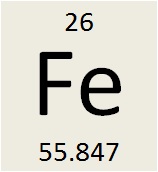7.4 - Atomic Symbols

The boxes on the periodic table of the elements tell you more than you would think about individual elements. From the boxes on the table, you instantly know how many protons, neutrons and electrons a neutral atom has. By its symbol, you know its name. You should not have to memorize the periodic table. You will be using it enough that by the end of the term you will pretty much know at least the first twenty elements well.
On the periodic table, three pieces of information are given to you. Let’s take iron as our example. If you look on your periodic table you may see something that looks like this:
The symbol, Fe, is the accepted symbol for iron. Many of the symbols for elements will make sense to you because they are abbreviations for the English name of that element. Examples are hydrogen (H), helium (He), oxygen (O), and neon (Ne). Other elements symbols may come from another language. Tungsten is (W) because in German it was called Wolfram. Many symbols come from the Latin name for the element. Gold (Au) is Auric. Lead (Pb) is Plumbum while silver (Ag) is Argentums. Whenever possible, you will be given both the symbol and the name of the element.
The number near the top of the box is called the atomic number. The atomic number tells you the number of protons that are in the nucleus of the atom. The number of protons determines what element you have. The number of neutrons and/or the number of electrons can change in an element. If the number of protons changes, it is no longer that element. Iron has 26 protons in its nucleus. If an atom has 25 protons it can only be manganese while 27 protons can only make up cobalt. Iron, and only iron, has 26 protons in its nucleus.
The number near the top of the box is called the atomic number. The atomic number tells you the number of protons that are in the nucleus of the atom. The number of protons determines what element you have. The number of neutrons and/or the number of electrons can change in an element. If the number of protons changes, it is no longer that element. Iron has 26 protons in its nucleus. If an atom has 25 protons it can only be manganese while 27 protons can only make up cobalt. Iron, and only iron, has 26 protons in its nucleus.
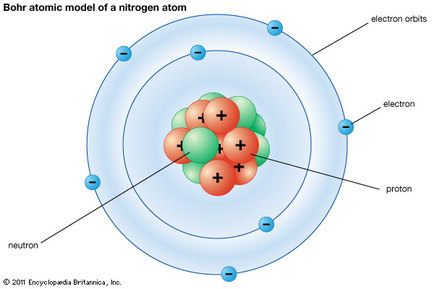
Atoms, when alone are considered electrically neutral. This means that there is no positive or negative charge on the entire atom. Since protons are positively charged, there must be an equal number of electrons (negative charges) in a neutral atom. This means that if iron has 26 protons (atomic number 26), its neutral state would also have 26 electrons.
The number at the bottom-right of the symbol, 55.847, is called the atomic mass. (Some textbooks also use the term atomic weight). The atomic mass is the mass of the nucleus of the atom. The mass of the electrons is not added to the atomic mass. Why? The mass of the electron is 1/2000 the mass of a proton so it is negligible compared to the mass of a proton.
Both the proton and the neutron have an atomic mass of 1. If that is true, how could iron have a total mass of 55.847? The answer is somewhat involved but the short answer is that the atomic mass given on the periodic table tells you more about the atom than just the number of protons and neutrons. We will discuss the decimal part of the number when we get to isotopes. Right now, all you need do is round off the atomic mass to the nearest whole number.
By rounding the atomic mass of iron to 56, you can tell a more about the atom. By knowing its atomic number, you know the number of protons. The atomic mass tells you the mass of the nucleus. Since the nucleus is made up of protons and neutrons, the difference between the atomic mass and the atomic number is the number of neutrons in the atom!
The number at the bottom-right of the symbol, 55.847, is called the atomic mass. (Some textbooks also use the term atomic weight). The atomic mass is the mass of the nucleus of the atom. The mass of the electrons is not added to the atomic mass. Why? The mass of the electron is 1/2000 the mass of a proton so it is negligible compared to the mass of a proton.
Both the proton and the neutron have an atomic mass of 1. If that is true, how could iron have a total mass of 55.847? The answer is somewhat involved but the short answer is that the atomic mass given on the periodic table tells you more about the atom than just the number of protons and neutrons. We will discuss the decimal part of the number when we get to isotopes. Right now, all you need do is round off the atomic mass to the nearest whole number.
By rounding the atomic mass of iron to 56, you can tell a more about the atom. By knowing its atomic number, you know the number of protons. The atomic mass tells you the mass of the nucleus. Since the nucleus is made up of protons and neutrons, the difference between the atomic mass and the atomic number is the number of neutrons in the atom!
7.4 Example Question 1
7.4 Example Question 2
7.4 Review Questions
| 7.4_review_questions.pdf | |
| File Size: | 101 kb |
| File Type: | |